Electronic Products first reported the development of a self-charging battery in 2012, when developers at Georgia Institute of Technology began putting their plans into action. Professor Zhong Lin Wang led his team in creating the world’s premiere self-charging power pack that enabled charging without being plugged into any external source of electricity like a wall socket. The battery charged when mechanical stress was applied to the device, forcing lithium ions to move from the cathode to the anode, allowing the piezoelectric effect to ensue. Since 2012, the scientists have honed their skills to create a new and improved battery that adds nanoparticles to the battery’s piezoelectric material. This addition completely upgrades the battery, resulting in a more efficient charge and higher capacity for storage.
Recently, Wang corralled researchers from Lanzhou University, Northeasteren University in Shenyang, and the Chinese Academy of Sciences in Beijing to coauthor a paper that was published in the science journal Nanotechnology .
The battery itself is impressive. It is hundreds of micrometers thick, and is small enough to fit inside a steel coin-like cell. For example, if this battery were placed under a calculator’s buttons, the pressure from the user’s fingers would generate mechanical power. This mechanical energy can be converted into chemical energy and stored in the battery. Zhang believes that this technology will eventually be integrated into self-powered sensor systems and portable electronics like cell phones and devices like heart monitors.
The battery’s ability to self-charge, convert, and store energy is truly remarkable. Its abilities differentiate its performance from regular batteries whose processes include energy conversion that occurs in a separate device. This new battery skips over the middle step of energy conversion to electricity. This process is highly efficient and more dynamic than using two separate devices.
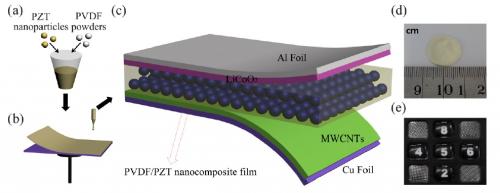
The scientific team at the Georgia Institute of Technology changed a Li-ion battery into one that charged itself by exchanging the polyethylene separator, splitting the two electrodes in a Li-ion battery and inserting a piezoelectric material. This created a charge when pressure was applied. Since 2012, the researchers have enhanced the use of PVDF film by including a lead zirconate titanate (PZT) nanoparticle in the PVDF film. Here, a nanocomposite was created.
PZT’s presence in the battery has made its activity more efficient, increasing the storage capacity 2.5 times more than the previous edition; this battery’s storage has increased from 0.004 to 0.010 μA/h. These significant changes show how a nanocomposite film can be beneficial to the overall performance of self-charging batteries. The team of researchers plans to expound their development efforts in the future.
Story via Electronic Products, Phys.org
Advertisement
Learn more about Electronic Products Magazine





